Chemistry
Polymer physics, polymer chemistry and chemical technology related to Avans Hogeschool (Hogeschool Brabant), Faculteit Techniek & Natuur, Breda

Examination Papers
The following examples of preliminary examination papers in Dutch come on students' requests.
Chemische Procestechniek ![]()
Polymeren I ![]()
Polymeren II ![]()
You need Adobe Acrobat Reader to view the documents above. A free copy of Acrobat Reader is downloadable from Adobe's web site.
![]()
Supporting Calculator Software
Burgers' Creep
In what way will a sample of a viscoelastic material like an amorphous polymer elongate in course of time, if it is subjected to a constant stress? Based on the Burgers model the mechanical behavior is given numerically as well as graphically. Read more...
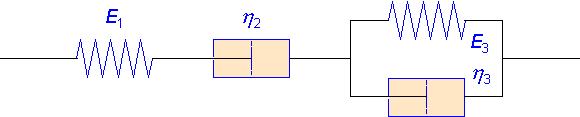
Burgers' Stress Relaxation
A viscoelastic material like an amorphous polymer is stretched to a given value of the strain. How does the stress necessary to maintain the constant strain, changes as a function of time? Grounded on the Burgers model, the question is numerically and graphically answered.
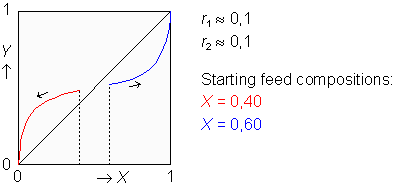
Copolymer Composition Drift
During the synthesis of a bipolymer, the composition of the instantaneously formed copolymer chains usually changes as the polymerization reaction proceeds. The changing composition is plotted as a function of the also changing composition of the monomer feed, starting from the initial feed composition and depending upon two given reactivity ratios. Read more...
Rectification (Distillate Composition Calculation)
According to the McCabe-Thiele method the program calculates the composition of the distillate obtained from a two component liquid mixture with known composition and relative volatility, boiling under a column with a given number of theoretical plates and a given reflux ratio. Read more...
Rectification (Calculation of the Number of Plates)
This program can be used to determine the total number of theoretical plates in a batch rectification unit, according to McCabe-Thiele. It needs input values of the relative volatility, the composition of the boiling mixture, the reflux ratio and the distillate composition. Read more...
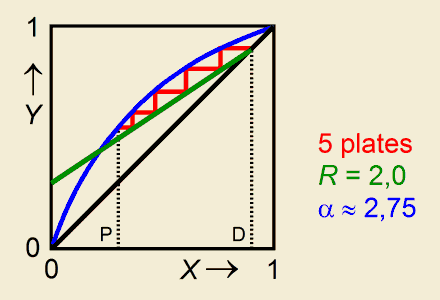
As is evident from many experiments, like the membrane osmometric determination of the number average molecular weight of a polymer, or the determination of the viscosity average molecular weight according to Mark-Houwink, a regression program offers in more respects a splendid relief in many working-outs. You can find regression programs elsewhere on this web site.
Models of Matter
Atomic orbitals
 |
 |
 |
(..) |
s (sharp) |
p (principal) |
d (diffuse) |
f (fundamental) |
 orbitals of neon (1s, 2s, 2p) |
|||
Some hybrid atomic orbitals
 |
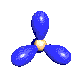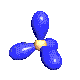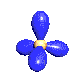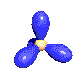 |
 |
 |
| sp² in ethylene, borane (BH3) |
sp³ polyethylene, ammonia (NH3) |
sp³d in PCl5, I3– (central I) |
sp³d² in SF6 |
Three-dimensional representation of a molecular model
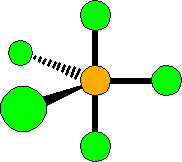
sp³d hybridization in a PCl5 molecule
A space-filling model of a dendrimer

(Dendrimers are repeatedly branched molecules with precise, nano-sized composition and well-defined three-dimensional shapes.)
Models of DNA (Crick and Watson's double helix, 1953)

Line-angle formula of chlorophyll a
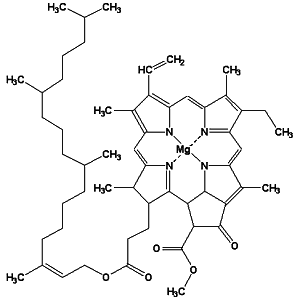
The line-angle formula is also called a skeletal structure, or a stick figure.

(Using red and blue light absorbed from the sunlight chlorophyll enables plants to synthesize carbohydrates, thus converting light energy into chemical energy.)
Full structural formula of polyurethanes (PU)
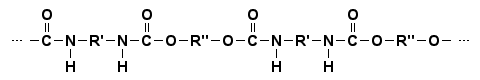
In a full (or expanded) structural formula all the individual chemical bonds should be visible. In the general polyurethane formula, however, the groups R' and R" haven't been written out, since their structures depend on the many distinguishable polyurethane types. If, for instance, the polymer is made from the monomers hexamethylenediisocyanate and ethylene glycol only, the groups R' and R" are (CH2)6 and (CH2)2 respectively. The following structures show the repeating dyad in this particular polymer chain.
Full structural formula
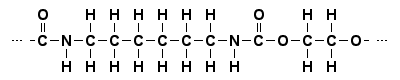
Condensed structural formula
...CONH(CH2)6NHCOO(CH2)2O... or
...-CO-NH-(CH2)6-NH-CO-O-(CH2)2-O-...
Ball-and-stick model
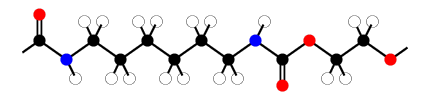
Lewis structure
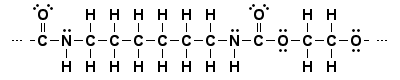
Space-filling models
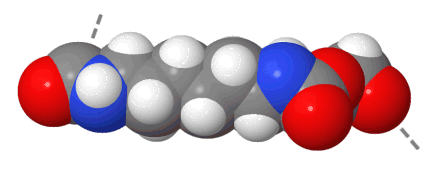
Space is filled up to 100 % of the van der Waals radius
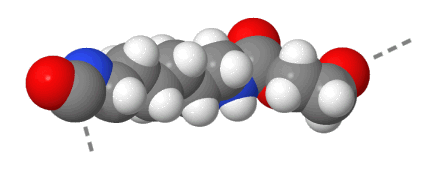
Space-filling: 75 % of the van der Waals radius
Both space-filling models are drawn to the same scale, but from a different viewing angle. In a real chain the distance between both ends of a repeating unit is continuously changing and roughly amounts to 1,5 nm.
Some polyurethane containing products
(move mouse cursor over pictures)